"When I want to eliminate patient post-op visits for suture removal, I have been using the HEMIGARD device with the retention suture, followed by deeps and top fast gut sutures." Stephanie Howerter, DO, FACMS
With the recent need for physical distancing, there has been a renewed interest in closure methods that do not require removal by the provider. When closing wounds, there is usually an absorbable “deep” or dermal layer of sutures or staples combined with a “top” or superficial layer of sutures, staples or adhesives. For many excisional wounds, there is significant tension, especially in the middle of the wound. In these cases, if the dermal absorbable sutures were to prematurely fail, then all of the tension would be borne by the top sutures. Let’s review the options for the top layer of a wound closure:
- Nonabsorbable sutures such as nylon and polypropylene need to be removed, usually within 6-8 weeks. For many reasons, heightened by the COVID-19 pandemic, avoiding these nonabsorbable suture removals is advisable. Aside from protecting staff and patients from COVID-19 exposure, patient convenience and reduction of staffing needed to remove sutures are other benefits.
- Nonabsorbable staples. Stainless steel staples are somewhat easier for patients to remove, because they or a family member can be given a staple remover. Many surgeons like using staples for scalp, but less so for the fragile skin of many other body sites.
- Absorbable sutures like fast absorbing gut can be used for the top layer. The disadvantage of this suture is that it dries quickly and become brittle. After several days, these may fail spontaneously, adding little or no benefit to the absorbable dermal suture. For wound closures under minimal tension (like a skin graft), these work fine and allow the patient to avoid a follow-up visit.
- Glue alone. For some wounds under low tension that have been meticulously closed with dermal sutures, the superficial closure may be done with only glue. However, if the glue were to prematurely fail (e.g. get wet), then it could lead to wound closure failure (dehiscence).
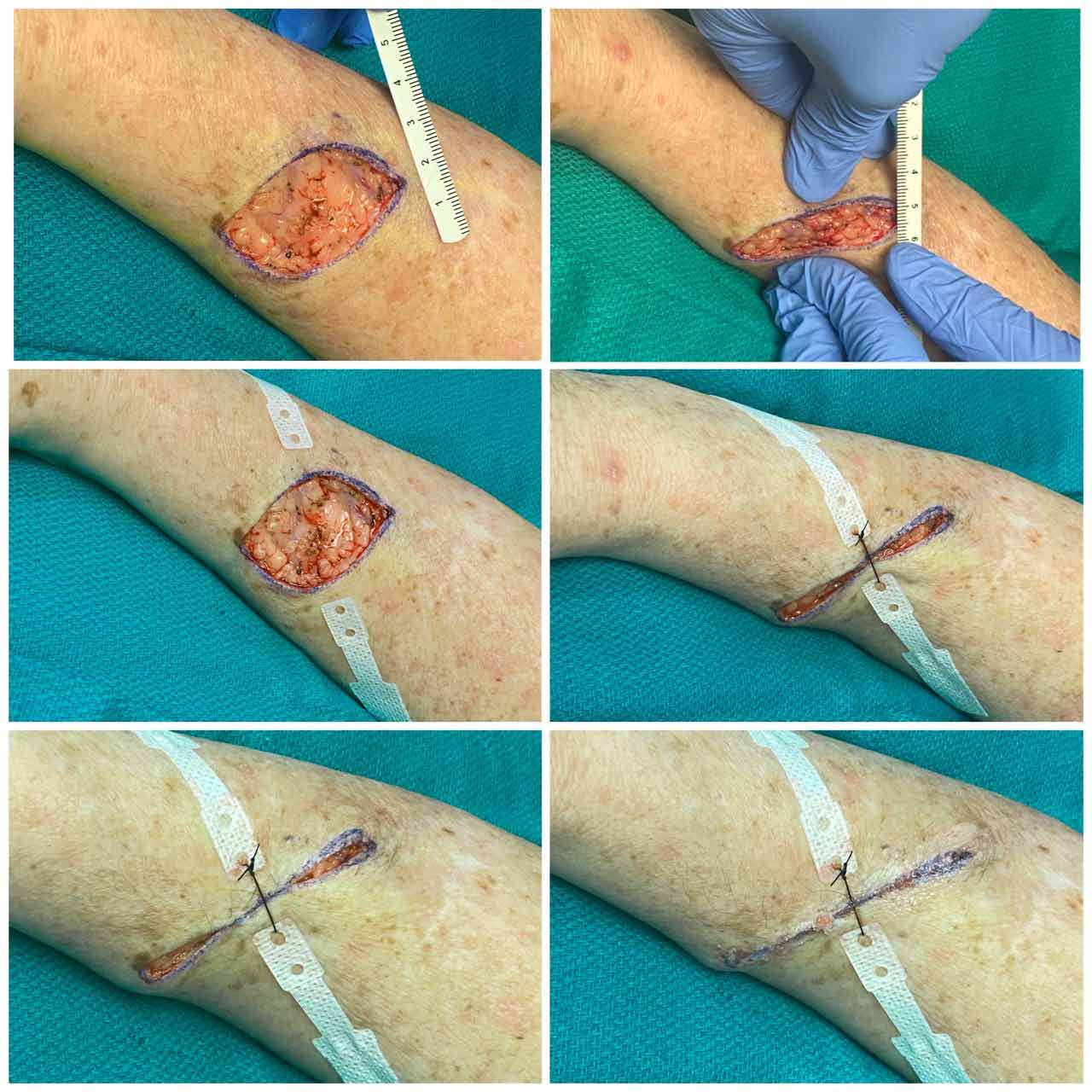
- HEMIGARD® ARS device. Prior to COVID-19, many patients underwent a closure with our new HEMIGARD® ARS device. In all of these cases, we used the HEMIGARD® ARS device and large caliber (e.g. 2-0 nylon) retention suture to relieve the wound tension in the middle of the elliptical wound. Absorbable dermal sutures (usually polyglactin) were used for dermal support. Because the HEMIGARD® device and accompanying retention suture bears the vast majority of the wound tension, we use either fast absorbing gut suture or glue (e.g. GluSeal®liquid bandage by GluStitch® Inc.) for remainder of wound closure.
Four steps to HEMIGARD® closure: ADHERE strips, RETENTION SUTURE in middle, DERMAL sutures, GLUE (or fast-absorbing gut sutures) on top
For wound care, the patient is to keep the HEMIGARD® strips dry. The use of GluSeal® liquid bandage for superficial closure both seals the wound and reduces the need for applying ointments (which are not recommended).
To remove the HEMIGARD® ARS device, the patient simply:
- Cuts and removes the retention suture. The HEMIGARD® ARS device elevates the retention suture above the skin to allow patient to easily get scissors under the suture to cut it.
- Peels back the HEMIGARD strips. The patient grips the rigid end of each HEMIGARD® device and pulls AWAY from wound.
In the above images (in which the backgrounds were removed with post-processing), the patient was guided on removal of the suture and HEMIGARD® ARS device in their own home using a telemedicine portal. The HEMIGARD® ARS device gives providers another option for closing challenging wounds without the need for a suture removal follow-up visit.
It is the responsibility of each provider to understand and comply with local, state and federal orders regarding performance of medical procedures. Providers should also understand relevant guidance from the CDC and CMS. For skin cancers management during this pandemic, other good sources of guidance are available from the AAD, ACMS and NCCN. It is the responsibility of each provider to determine the most appropriate course of action for their tumors during and after this pandemic.